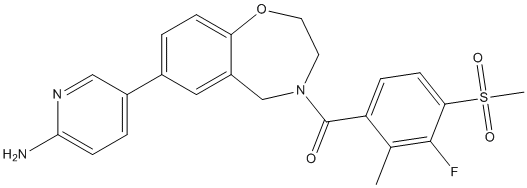
Bilastine
- Catalog NO.:A856214
- CAS No. : 202189-78-4
- Molecular Formula:C28H37N3O3
- Molecular Weight: 463.61
1. Please click the
 to confirm the discount and stock quantity.
to confirm the discount and stock quantity.2. Click“Bulk Inquiry”to ask for quotations of other quantity.
3. The price does not include shipping and bank charges.
Product Details
| CAS Number | 202189-78-4 | Appearance | white crystalline powder |
| Catalog Number | A856214 | MDL Number | MFCD09837814 |
| Molecular Formula | C28H37N3O3 | Molecular Weight | 463.61 |
| Boiling Point | 639.1±55.0°C at 760 mmHg | Melting Point | |
| Flash Point | Density | ||
| Synonyms | 2-[4-[2-[4-[1-(2-ethoxyethyl)benzoimidazol-2-yl]-1-piperidyl]ethyl]phenyl]-2-methyl-propanoic acid | ||
| Storage Condition | Store in a tightly closed container,in a cool and dry place | ||
Safety
| GHS Pictogram |

|
||
| Signal Word | Warning | UN Number | |
| Hazard Statements | H302 | Class | |
| Precautionary Statements | P280-P305+P351+P338 | Packing Group | |
Description
|
Bilastine (trade name Bilaxten) is a second generation antihistamine drug for the treatment of allergic rhinoconjunctivitis and urticaria (hives). It exerts its effect as a selective histamine H1 receptor antagonist, and has a potency similar to cetirizine and is superior to fexofenadine. It was developed in Spain by FAES Farma. Bilastine is approved in the European Union for the symptomatic treatment of allergic rhinoconjunctivitis and urticaria, but it is not approved by the U.S. Food and Drug Administration for any use in the United States. Bilastine has been effective in the treatment of ocular symptoms and diseases of allergies, including rhinoconjuctivitis. Additionally, bilastine has been shown to improve quality of life, and all nasal and ocular symptoms related to allergic rhinitis. |