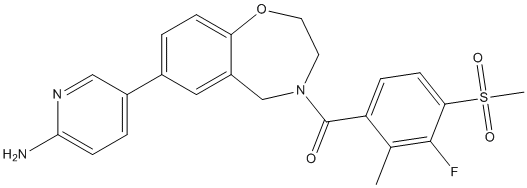
Macitentan
- Catalog NO.:A854671
- CAS No. : 441798-33-0
- Molecular Formula:C19H20Br2N6O4S
- Molecular Weight: 585.96
Note*
1. Please click the to confirm the discount and stock quantity.
to confirm the discount and stock quantity.
2. Click“Bulk Inquiry”to ask for quotations of other quantity.
3. The price does not include shipping and bank charges.
Bulk Inquiry
1. Please click the
 to confirm the discount and stock quantity.
to confirm the discount and stock quantity.2. Click“Bulk Inquiry”to ask for quotations of other quantity.
3. The price does not include shipping and bank charges.
Quality Control&SDS
Product Details
| CAS Number | 441798-33-0 | Appearance | |
| Catalog Number | A854671 | MDL Number | MFCD17167076 |
| Molecular Formula | C19H20Br2N6O4S | Molecular Weight | 585.96 |
| Boiling Point | 692.397°C at 760 mmHg | Melting Point | |
| Flash Point | Density | ||
| Synonyms | N-[5-(4-Bromophenyl)-6-[2-[(5-bromo-2-pyrimidinyl)oxy]ethoxy]-4-pyrimidinyl]-N'-propylsulfamide | ||
| Storage Condition | Store in a tightly closed container,in a cool and dry place | ||
Safety
| GHS Pictogram |

|
||
| Signal Word | Warning | UN Number | |
| Hazard Statements | H302 | Class | |
| Precautionary Statements | P280-P305+P351+P338 | Packing Group | |
Description
|
Macitentan (trade name Opsumit) is an endothelin receptor antagonist (ERA) approved for the treatment of pulmonary arterial hypertension (PAH). The other two ERAs marketed as of 2014 are bosentan and ambrisentan. Macitentan is a dual ERA, meaning that it acts as an antagonist of two endothelin (ET) receptor subtypes, ETA and ETB. However, macitentan has a 50-fold increased selectivity for the ETA subtype compared to the ETB subtype. The drug received approval from the U.S. Food and Drug Administration (FDA) on October 13, 2013. |