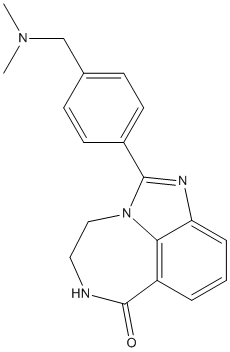
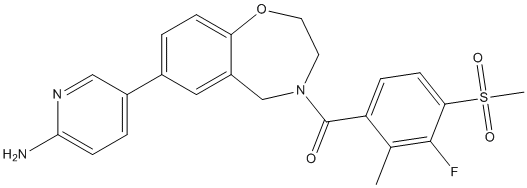
Conivaptan
- Catalog NO.:A879299
- CAS No. : 210101-16-9
- Molecular Formula:C32H26N4O2
- Molecular Weight: 498.57
1. Please click the
 to confirm the discount and stock quantity.
to confirm the discount and stock quantity.2. Click“Bulk Inquiry”to ask for quotations of other quantity.
3. The price does not include shipping and bank charges.
Product Details
| CAS Number | 210101-16-9 | Appearance | |
| Catalog Number | A879299 | MDL Number | |
| Molecular Formula | C32H26N4O2 | Molecular Weight | 498.57 |
| Boiling Point | Melting Point | ||
| Flash Point | Density | ||
| Synonyms | YM087; Conivaptan;N-[4-(2-methyl-4,5-dihydro-3H-imidazo[4,5-d][1]benzazepine-6-carbonyl)phenyl]-2-phenylbenzamide | ||
| Storage Condition | Store in a tightly closed container,in a cool and dry place | ||
Safety
| GHS Pictogram |

|
||
| Signal Word | Warning | UN Number | |
| Hazard Statements | Class | ||
| Precautionary Statements | Packing Group | ||
Description
|
Conivaptan (YM 087, brand name Vaprisol) is a non-peptide inhibitor of antidiuretic hormone (vasopressin receptor antagonist). It was approved in 2004 for hyponatremia (low blood sodium levels) caused by syndrome of inappropriate antidiuretic hormone (SIADH), and there is some evidence it may be effective in heart failure. It is marketed by Cumberland Pharmaceuticals, Inc. Conivaptan inhibits two of the three subtypes of the vasopressin receptor (V1a and V2). Effectively, it causes iatrogenic nephrogenic diabetes insipidus. Conivaptan has not been approved by the FDA for the treatment of decompensated congestive heart failure. However, in theory, vasopressin receptor antagonism would be particularly useful in this setting, and an initial study shows that it has some promise. |